How to Understand Catalytic Reactions and Their Importance?
Catalytic reactions play a crucial role in various chemical processes. These reactions involve catalysts that increase reaction rates without being consumed. The global catalyst market is projected to reach $38 billion by 2025, according to recent industry reports. This growth highlights the importance of catalytic reactions in sectors like petrochemicals and pharmaceuticals.
Understanding catalytic reactions can illuminate their significance in reducing energy consumption and minimizing waste. Many industrial processes depend on them for efficiency. For instance, catalytic converters in vehicles help reduce harmful emissions. This exemplifies how catalytic reactions can have direct environmental impacts.
However, not all catalytic processes are well understood. Some mechanisms remain elusive, leading to inefficiencies. Research is ongoing to optimize these reactions. A deeper understanding could further enhance production processes and environmental sustainability. We must reflect on the challenges ahead as the field evolves.

Understanding the Basics of Catalytic Reactions in Chemistry
Catalytic reactions play a vital role in chemistry. Catalysts accelerate chemical reactions without being consumed. They are present in small amounts but have a significant impact. Understanding these processes can enhance both industrial applications and research.
In many reactions, catalysts lower the activation energy. This makes it easier for reactions to occur under milder conditions. Many catalysts are metals, while others are enzymes. Each type has unique properties and functions. Not every catalyst works effectively in all reactions. It’s crucial to choose the right one based on the desired outcome.
Tips: Choose a catalyst that matches your reaction type. Consider reaction conditions such as temperature and pressure. Don’t overlook the impact of impurities in your reactions; they can hinder performance. Experimentation can lead to unexpected results, so be prepared to adapt. Understanding these principles is key to mastering catalytic reactions in chemistry.
Types of Catalysts: Homogeneous vs. Heterogeneous Catalysis
Catalysts play a crucial role in speeding up chemical reactions. When we talk about catalysts, we often categorize them into two types: homogeneous and heterogeneous.
Homogeneous catalysts exist in the same phase as the reactants. This means if the reactants are in a liquid state, the catalyst is also liquid. An example is the use of acid in esterification reactions. The catalyst blends well, allowing for efficient interaction with the reactants.
Heterogeneous catalysis, on the other hand, involves catalysts in a different phase than the reactants. This often means solids acting on gases or liquids. A common instance is the use of metal catalysts in the Haber process for ammonia production. The solid metal provides surface area for reactions to occur.
However, maintaining the efficiency of heterogeneous catalysts can be challenging. Factors like temperature and surface fouling can hinder performance. Addressing these issues may lead to advancements in catalyst technology but requires careful consideration and ongoing study.
The Mechanism of Catalytic Reactions: Key Steps and Intermediates
Catalytic reactions play a crucial role in chemical processes, influencing speed and efficiency. Understanding their mechanism involves key steps and intermediates that define the reaction pathway. A recent industry report indicates that over 80% of all chemical processes utilize catalysts. This statistic underscores the significance of catalysts in various sectors, including pharmaceuticals and petrochemicals.
At the core of catalytic reactions, intermediates are transient species formed during the process. These intermediates dictate the rate at which reactants transform into products. For instance, in hydrogenation reactions, metal catalysts form the key complexes that stabilize these intermediates. Studies show that optimizing these steps can enhance yield by 30% or more. However, the complexity of these reactions often leads to unforeseen challenges, such as catalyst deactivation or selectivity issues.
Research continues to evolve in this area. Yet, many catalytic systems are still poorly understood. The intricate balance of reaction conditions, such as temperature and pressure, can dramatically alter the reaction outcome. Recent analysis suggests that up to 25% of industrial catalysts operate below their potential efficiency. This gap highlights the need for further exploration of catalytic mechanisms to fully harness their capabilities.
Importance of Catalysis in Industrial Processes and Economic Impact

Catalysis plays a crucial role in various industrial processes, significantly shaping economic outcomes. According to a report by the International Energy Agency, catalysts are involved in about 90% of all chemical processes. This highlights their essential function in enhancing reaction rates and reducing energy consumption. For instance, the Haber-Bosch process, used for ammonia synthesis, relies on catalysts to boost production efficiency, impacting agriculture and food supply globally.
The economic implications of catalysis are noteworthy. A study from the World Catalysis Association estimates that catalysis contributes over $3 trillion to the global economy annually. This figure underscores the importance of efficiently designed catalysts that can minimize production costs and maximize output. Yet, despite these advances, there are ongoing challenges. Many catalysts still suffer from deactivation over time, which influences long-term sustainability. Efforts to develop more robust catalysts are crucial but often fall short in practical applications, necessitating further innovation.
As industries strive for greener alternatives, the need for advanced catalytic technologies is pressing. Transitioning toward more sustainable and efficient processes can significantly lower greenhouse gas emissions. Yet, without comprehensive research and development, these transitions may remain stagnant. Balancing economic growth and environmental responsibility through effective catalysis will be a continuous journey.
Applications of Catalytic Reactions in Environmental Sustainability and Energy
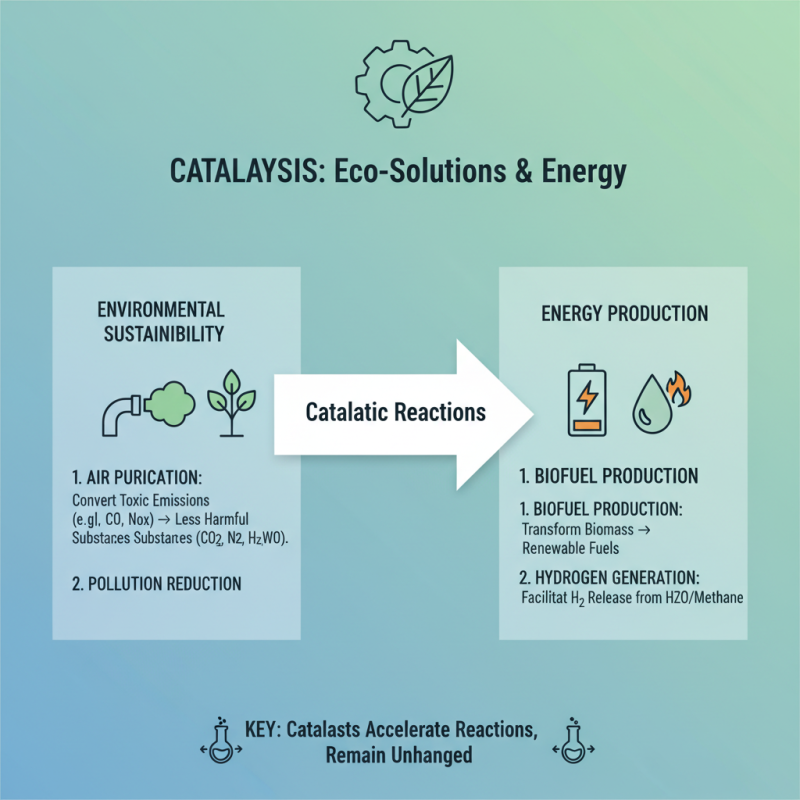
Catalytic reactions play a crucial role in environmental sustainability and energy production. These processes involve catalysts that speed up reactions without being consumed. For instance, they are vital in converting harmful emissions into less toxic forms. This transformation is crucial for reducing air pollution and improving air quality.
One significant application is in the production of cleaner fuels. Catalysts help refine crude oil into gasoline, making it more efficient. They also facilitate renewable energy processes, such as hydrogen production through water splitting. However, the efficiency of these reactions can vary. Sometimes they do not achieve the desired yield. This unpredictability raises questions about the optimization of catalysts.
Furthermore, catalytic reactions can contribute to waste management strategies. They can assist in breaking down harmful waste products effectively. Yet, it's essential to continually research and innovate. We need to identify better catalysts that are more environmentally friendly. This ongoing journey invites a deeper reflection on our methods and improvements. The balance between efficiency and sustainability is delicate, demanding constant attention and effort.
Related Posts
-
What is a Catalyst in Catalysis? Understanding Their Role and Impact in Chemical Reactions
-

Top 10 Types of Chemical Catalysts You Should Know?
-

How to Accelerate Your Catalysis Research for Breakthrough Discoveries
-
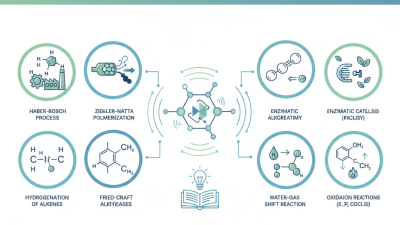
Top 10 Catalytic Reactions You Should Know For Chemistry Success
-

Why Do Catalysts Matter in Chemistry and How Do They Work
-

Top 10 Best Catalytic Reactions You Need to Know?
