Top 10 Most Effective Catalysts in Chemical Reactions?
In the ever-evolving world of chemistry, catalysts play a pivotal role in enhancing reaction efficiency. According to a recent report by the International Society for Catalysis Research, catalysts can improve reaction rates by up to 100 times. Among the numerous types available, the "4 catalyst" category stands out for its effectiveness in various applications. Experts agree that understanding these catalysts can lead to significant advancements.
Dr. Emma Carr, a leading figure in catalysis research, states, “The right catalyst can transform industries.” This powerful insight underlines how critical it is to explore the top catalysts available. Visualizing this impact is essential. For instance, in the production of ammonia, a well-chosen catalyst can reduce energy consumption dramatically, showcasing both efficiency and sustainability.
However, the field is not without challenges. Some commonly used catalysts may face performance limitations over time. This raises questions about their longevity and effectiveness. Continuous research is necessary to address these gaps and improve the overall efficiency of catalytic processes. Understanding the nuances of "4 catalyst" options will be key to future innovations.

Definition and Role of Catalysts in Chemical Reactions
Catalysts play a critical role in chemical reactions. They are substances that increase the reaction rate without being consumed in the process. This unique property makes them invaluable in various industrial applications. Catalysts often lower the activation energy required for reactions, allowing them to happen more efficiently.
In catalysis, there are different types. Homogeneous catalysts mix with the reactants, while heterogeneous catalysts exist in a different phase. Each type has its pros and cons. For instance, heterogeneous catalysts can be easier to separate from products. However, they may also show lower selectivity.
Here are some tips for effective catalysis. Choose the right catalyst based on your reaction type. Always consider the reaction conditions. Temperature and pressure can significantly impact catalyst effectiveness. Also, remember that not all catalysts work perfectly in every situation.
Continuously test and adjust. Sometimes, a catalyst may lose efficiency over time due to factors like poisoning or sintering. Regular maintenance and monitoring are essential.
Overview of Factors Influencing Catalyst Effectiveness
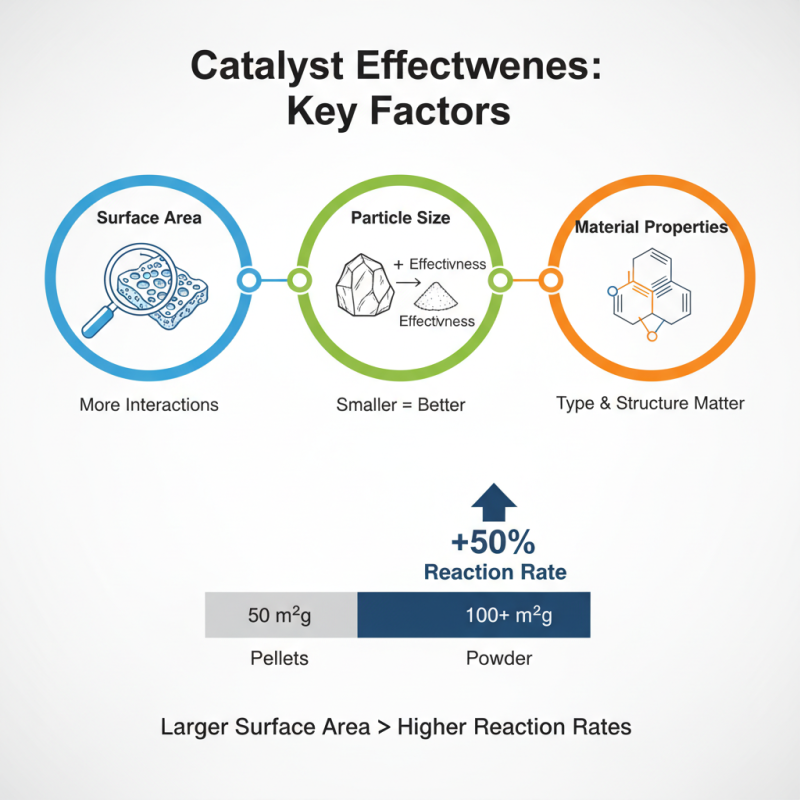
Catalyst effectiveness in chemical reactions relies on multiple influencing factors. The surface area is crucial. A larger surface area allows more reactant interactions. For instance, powdered catalysts often outperform larger pellets. In studies, catalysts with a surface area over 100 m²/g showed up to 50% higher reaction rates. This highlights the role of physical properties in catalysis.
Another key factor is the reaction environment. Temperature and pressure significantly impact catalyst performance. Higher temperatures can increase reaction rates but may lead to catalyst degradation. Additionally, optimal pressure levels can enhance reaction efficiency. Research indicates that reactions conducted at elevated pressures can increase yields by 30-40%. However, these conditions must be balanced to avoid compromising catalyst integrity.
Lastly, the chemical composition is vital. Different metal combinations exhibit unique catalytic properties. For example, an alloy may show improved strength but could be less active than a pure metal. This presents a challenge, as one must consider trade-offs. There's also the issue of catalyst poisoning, where contaminants reduce effectiveness. While advancements are ongoing, these factors necessitate continuous examination and innovation in catalyst development.
Top 10 Most Effective Catalysts and Their Applications
Catalysts play a crucial role in speeding up chemical reactions. They reduce the energy required for these processes. For instance, platinum is an effective catalyst in automotive catalytic converters. This technology helps in reducing harmful emissions. The Global Catalysis Market Report states that the market is expected to reach $38 billion by 2025. This growth reflects the rising demand for cleaner production methods.
In the pharmaceutical industry, catalysts enhance drug development efficiency. They can enable reactions that create complex molecules swiftly. A report from the American Chemical Society highlights that 70% of modern drugs rely on catalytic processes. However, many efficient catalysts are expensive and sometimes difficult to recycle. The challenge lies in balancing cost-effectiveness with performance.
Additionally, bio-catalysts have emerged as an eco-friendly alternative. Enzymes and microorganisms can catalyze reactions under mild conditions. Studies show they help reduce energy usage by over 30%. Yet, further research is needed to improve their stability and applicability in industrial settings. There’s a long way to go before achieving widespread adoption of these innovative catalysts.
Comparison of Heterogeneous and Homogeneous Catalysts
Catalysts play a crucial role in chemical reactions. They can be categorized into two main types: homogeneous and heterogeneous. Homogeneous catalysts exist in the same phase as the reactants, usually in a solution. They are effective because they provide a uniform environment for reactions. However, separating them from the final product can be challenging. This often results in higher costs and potential impurity issues.
Heterogeneous catalysts, on the other hand, exist in a different phase. Typically, they are solid while the reactants are gases or liquids. This phase difference allows for easier separation. For example, in catalytic converters, solid catalysts facilitate gas reactions, making the process efficient. Yet, heterogeneous catalysts may suffer from issues like limited surface area and deactivation over time. Not all reactions work well with them.
Understanding the strengths and weaknesses of each catalyst type is vital. Selecting the right catalyst can enhance reaction rates and product yields. Each has its unique advantages and potential drawbacks. Experiments often reveal unexpected outcomes. Refinement in catalyst selection could lead to more efficient processes in various industries.
Future Trends in Catalyst Development and Research

The development of catalysts is evolving rapidly. Researchers are focusing on nanomaterials and their unique properties. These tiny particles can enhance reactions at a lower energy cost. For instance, metal nanoparticles can offer increased surface area. This leads to more active sites for reactions, promoting efficiency. However, the stability of these materials must be improved. Sometimes they degrade quickly, which is a significant concern.
Sustainability is another important trend in catalyst research. Many scientists are investigating greener alternatives. Biocatalysts, derived from natural sources, show promise. They often operate under mild conditions and reduce harmful byproducts. Yet, their limited availability remains a challenge. Moreover, the requirement for specific conditions complicates their widespread use.
The quest for new catalytic pathways continues. Research is exploring new, underutilized reactions. These could lead to surprising efficiencies and novel products. However, the path is not always clear. Many potential catalysts show mixed results in practical applications. Iterative testing and refinement remain critical in advancing this field. The journey of catalyst development is filled with opportunities and obstacles alike.
Related Posts
-

How to Leverage Catalyst Technologies for Business Growth and Innovation
-
2026 Best Chemical Catalyst Innovations and Trends for Sustainable Solutions?
-
What is a Catalyst in Catalysis? Understanding Their Role and Impact in Chemical Reactions
-

How to Accelerate Your Catalysis Research for Breakthrough Discoveries
-
Top 10 Catalytic Reactions You Should Know For Chemistry Success
-

What Are the 4 Types of Catalysts and Their Functions in Chemistry?
