Why Are Catalytic Reactions Important in Chemical Processes?
Catalytic reactions play a crucial role in modern chemical processes. They enhance efficiency and selectivity, transforming raw materials into valuable products. This is vital for industries like pharmaceuticals and energy.
These reactions often proceed at lower temperatures and pressures, reducing energy usage. They also minimize waste, aligning with sustainable practices. However, not all catalytic reactions are perfect. Some may require extensive optimization, risking time and resources.
Despite their importance, challenges remain. Researchers strive to improve catalyst stability and selectivity. The intricate nature of catalytic mechanisms offers room for exploration. Understanding these reactions better can lead to groundbreaking advancements.
The Role of Catalytic Reactions in Enhancing Reaction Rates
Catalytic reactions play a crucial role in enhancing reaction rates. They enable chemical processes to occur faster and with less energy. This acceleration occurs without the catalyst being consumed. Catalysts change the reaction pathway, lowering the activation energy needed. This means more molecules can react at a given temperature.
For example, in the production of ammonia, catalysts make the process efficient. The Haber process, which creates ammonia from nitrogen and hydrogen gas, relies heavily on catalysts. This method drastically reduces the energy required and speeds up production. It’s a perfect illustration of how catalysts impact industrial processes.
**Tip:** Consider the temperature and pressure when using catalysts. These conditions can influence the effectiveness of the reactions greatly.
Creating a reaction with a catalyst often means trial and error. Not all catalysts work equally well for every reaction. Some may seem perfect but fail under certain conditions. Reflecting on these challenges is essential for improvement.
**Tip:** Conduct small-scale experiments to test different catalysts. This can help you find the best option for your specific needs.
Types of Catalysts Used in Chemical Processes
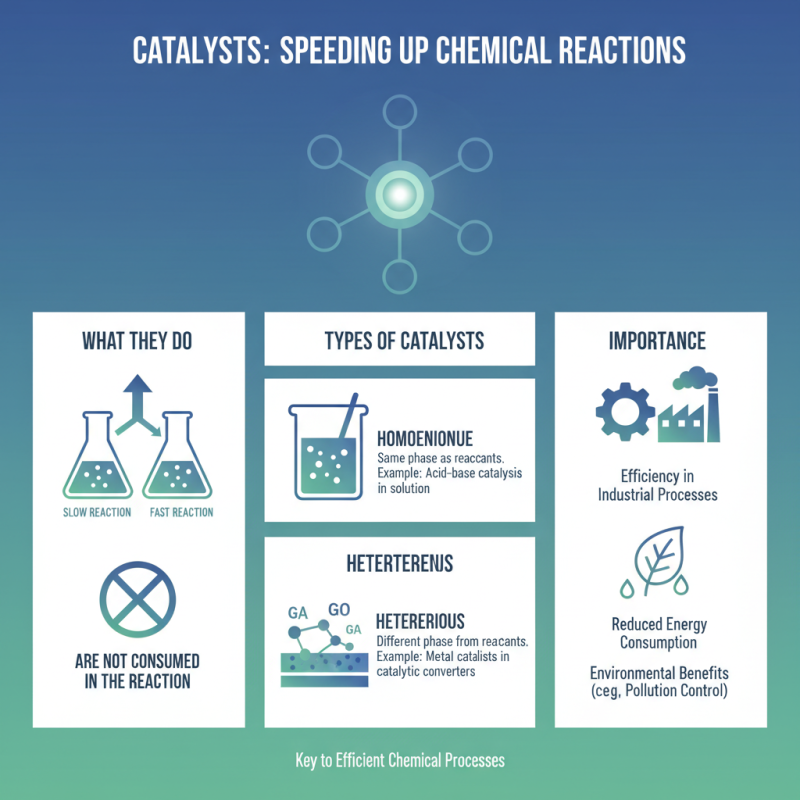
Catalysts play a vital role in chemical processes. They speed up reactions without being consumed. Understanding the types of catalysts is essential for designing efficient industrial processes.
There are two main categories of catalysts: homogeneous and heterogeneous. Homogeneous catalysts are in the same phase as the reactants. For example, they often exist in a liquid solution. These catalysts can provide unique advantages, but they may also create challenges for separation after the reaction.
On the other hand, heterogeneous catalysts are in a different phase than the reactants. They are typically solid while the reactants are gases or liquids. This type of catalyst is commonly used in processes like petroleum refining. While they allow easier separation, they may suffer from deactivation over time. Finding solutions to these challenges is ongoing. Each type of catalyst has its placing chemical industries, and the choice is not always straightforward.
Mechanisms of Catalytic Reactions and Their Efficiency
Catalytic reactions are crucial in enhancing the efficiency of chemical processes. They speed up reactions without being consumed. This characteristic helps in reducing energy consumption. The efficiency of catalysts can be influenced by factors like temperature and pressure. Industry reports indicate that catalysts can increase reaction rates by up to 100 times. This efficiency leads to significant cost reductions.
Understanding the mechanisms of catalytic reactions is key. These reactions often involve the formation and breaking of chemical bonds. In many processes, the catalyst lowers the activation energy required. For example, heterogeneous catalysts, which are in a different phase than the reactants, are widely used. They can offer higher surface areas for reactions, making them more effective.
**Tips:** When selecting a catalyst, consider its reusability. A catalyst that can be used multiple times saves money and resources. Keep in mind, not all catalysts work well for every reaction. Complex reactions sometimes require tailored solutions. Test different catalysts to find the best fit for specific needs. This trial-and-error approach can be enlightening but often leads to unexpected outcomes.
| Reaction Type | Catalyst Type | Mechanism | Efficiency (%) | Applications |
|---|---|---|---|---|
| Hydrogenation | Metal-based | Adsorption and desorption of reactants | 90 | Food industry, pharmaceuticals |
| Oxidation | Transition Metals | Electron transfer | 85 | Chemical synthesis, air pollution control |
| Dehydration | Acid catalysts | Protonation of substrate | 80 | Biofuel production, fine chemicals |
| Polymerization | Ziegler-Natta | Coordination mechanisms | 95 | Plastic manufacturing, textile industry |
| Cracking | Zeolite catalysts | Formation of free radicals | 88 | Fuels production, petrochemicals |
Impact of Catalysis on Industrial Chemical Production
Catalysis plays a critical role in industrial chemical production. It enhances reaction rates and improves efficiency. According to a report by the International Energy Agency, catalytic processes account for over 80% of all industrial chemical processes. This percentage highlights the significance of catalysis in large-scale manufacturing.
In many cases, catalysts help reduce energy consumption. For instance, the catalytic converters in automobiles lower harmful emissions. They enable cleaner combustion processes. However, the effectiveness of these catalysts often depends on complex variables, such as temperature and concentration. Sometimes, catalysts can even be deactivated by impurities.
Another surprising fact is that around 70% of chemical products rely on catalytic methods. This figure underscores the heavy reliance on catalytic processes in fields like pharmaceuticals and petrochemicals. Yet, challenges remain. Development costs for new catalysts can be high. Researchers continuously strive to improve catalyst design and reduce costs. The path forward involves innovative approaches and collaboration across disciplines.
Impact of Catalysis on Industrial Chemical Production
This chart illustrates the importance of catalytic reactions in various industrial chemical processes, highlighting the percentage contribution of catalyst usage in different sectors.
Environmental Benefits of Catalytic Reactions in Sustainability
Catalytic reactions play a critical role in making chemical processes more efficient. They lower the energy required for reactions, which can lead to less energy consumption overall. This efficiency is essential for reducing greenhouse gas emissions. When reactions occur more smoothly, we consume fewer resources and produce less waste.
Moreover, catalysts are vital for sustainable practices. They help convert raw materials into useful products without exhausting resources. For instance, in the production of biofuels, catalysts can streamline the conversion of organic materials. This process not only provides cleaner energy but also reduces dependence on fossil fuels. However, there are challenges. Not all catalytic processes are perfectly efficient, and some still produce harmful byproducts.
The environmental benefits of catalytic reactions are significant, yet not without flaws. We must continually improve these processes to maximize their positive impact. Research is ongoing, and while progress is made, the journey toward a sustainable future requires persistence and innovation. Balancing efficiency and sustainability is a goal worth striving for.
Related Posts
-

Top 10 Best Catalytic Reactions You Need to Know?
-

How to Accelerate Your Catalysis Research for Breakthrough Discoveries
-

What Are the 4 Types of Catalysts and Their Functions in Chemistry?
-

10 Essential Tips for Choosing the Right Catalysts and Technologies
-

How to Understand Catalytic Reactions and Their Importance?
-
What is a Catalyst in Catalysis? Understanding Their Role and Impact in Chemical Reactions
